THEORY
Continuous Electrodeionization (CEDI) is a process of removing ionized, (or ionizable), substances from water using ion exchange membranes, electrically active media, (ion exchange resin), and a DC potential.
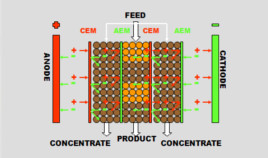
Feed water, (RO permeate), is feed through a series of alternating product and concentrate compartments with electrode compartments at either end. Both the product and reject compartments contain ion exchange resin. One product and one reject compartment is called a cell pair and each CEDI ‘module’ consists of multiple cell pairs.
Ion exchange resins in the dilute compartments accept ions in the feed water in a similar manner to conventional mixed bed ion exchange. However in CEDI, the ion exchange resin also serves as an ‘electrical bridge’ for the cations and anions to travel rather than the conventional method of ion exchange where they are ionically bound and require chemicals to be removed.
Membranes are essentially ion exchange resin manufactured in sheet form. They are selectively permeable to either anions or cations, depending on which type of resin the membrane is manufactured from, i.e., cation exchange membranes (CEMs), will allow only cations to pass through, and the anion exchange membranes, (AEMs), will allow only anions to pass through.
DC power is applied using a positive electrode (anode) and a negative electrode (cathode). As feed water enters each product compartment, anions (negatively charged ions) are attracted towards the anode through the selectively permeable anion exchange membrane . Similarly cations (positively charged ions) are attracted toward the cathode through the cation exchange membranes. Once in the concentrating compartment, the ions are carried away by the concentrate flow to drain, or in some cases is recycled.
Water passing through the product compartments becomes increasingly pure and, because DC voltage is applied continually, some of this purified water dissociates. Water molecules are dissociated, or broken down, into hydrogen and hydroxyl ions and this ‘acid’ and ‘caustic’ chemically regenerates the resin. This results in a module that continually regenerates itself during operation.
Electrochemical technology has continually improved. CEDI systems, operating in a single pass arrangement, can now purify water to levels approaching theoretical purity of 18.2 megohm-cm (0.055 microsiemens/cm). These performance improvements have been obtained through the years by a variety of means;
The use of the all filled designs, (resin in the dilute and concentrate compartments), and improvements to the electrode design have decreased the electrical resistance of modules. Reducing module resistance increases the available current for the voltage applied, and it is the current that is the ‘driving force’ in the process.
The choice and conditioning of the resins has continually improved. Resins can now be selected, conditioned and mixed for their water splitting capabilities.
Ion exchange membranes have been drastically improved and are now effectively impermeable to water, giving virtually no cross-leaks.
The design of CDI systems has also changed significantly, with standard designs incorporating materials of construction suitable for the Pharmaceutical and Microelectronics industries. In the power industry, where ‘size matters’, designs of up to 400m3/hr are, essentially, standard!
 |